- OT
- Industry
- Equipment and suppliers
- “I developed a drive to understand the retina in more detail”
Behind the brand
“I developed a drive to understand the retina in more detail”
Abdul Jahangir, director of Sense Medical, on the scope for AI and the importance of connectivity

24 October 2023
Three facts about Sense Medical
- Sense Medical was formed out of a UK subsidiary for the Japanese company, Kowa
- February 2023 marked the start of a distributorship for Canon Medical ophthalmic eye care products in the UK and Ireland
- Sense Medical is certified to ISO 13485 in Medical Devices Quality Management Systems.
Sense Medical – behind the scenes
Could you tell us how Sense Medical was formed?
The team at Sense Medical come from an optical background. In the course of my career, I developed a drive to understand the retina in more detail. Seeing prescriptions coming through for ophthalmic lenses and contact lenses is one element of providing better vision, but there could be a lot going on in the retina that could restrict a patient's vision. I moved from the optical field into practice management software in the very early days of paperless record systems, and then stepped from there into medical devices and imaging.
I was hired as head of international business for a Japanese pharmaceutical company, Kowa, which had a medical device arm in ophthalmology. Around that time, a grant had been given to optometrists in Scotland to purchase fundus cameras, and Kowa developed a large foothold in the market. I joined the company to leverage on that by launching a UK subsidiary in 2010.
In 2014, Kowa decided to centralise the business in Europe in order to launch a new product category in intraocular lenses. While I was invited to move with the business, I decided that I didn’t want to relocate, and so the senior management of Kowa offered to let me take over the UK business as a spin-off. This would take us from a subsidiary to an independent business for the UK operations. Sense Medical was registered as a business in 2014. I’m forever grateful to my colleagues in Japan for the advice and support in ensuring this independent business would be a success.
What is your focus as a company?
Sense Medical
was formed in 2014
This year, Sense Medical became the UK distributor for Canon Medical’s ophthalmic eye care products. How did this come about?
It’s a small world. In Japan, Kowa and Canon work closely as partners in ophthalmology. In 2020, when Kowa decided to shrink its imaging business, it began promoting Canon’s products in Japan for ophthalmology and recommended us to Canon in Europe. We wanted to form a close alliance and be a partner rather than solely a distributor, and so we co-branded, meaning both our logos will be on most of the marketing we do. We will not take on distributorships that would conflict with Canon Eyecare, because we signed in to be a flagship brand of Canon eye care in the UK and Ireland.
What is the latest product or service that you have introduced through Sense Medical?
The product we launched in September, the Xephilio OCT-R1, will revolutionise two key markets: diabetic eye screening, and High Street optometry. Over the last few years, Canon has developed an optical coherence tomography (OCT) device that can do everything in one smaller footprint device that is easy to use.
Optometry practices, from high end to mid-multiple and multiples, don’t have the resource to spend a lot of time training individuals. What is needed is a product that will take care of everything. Canon’s artificial intelligence (AI) technology is going to be a gamechanger in eye care. Utilising this technology to make clinical diagnoses, they will be able to spend more time with their patients.
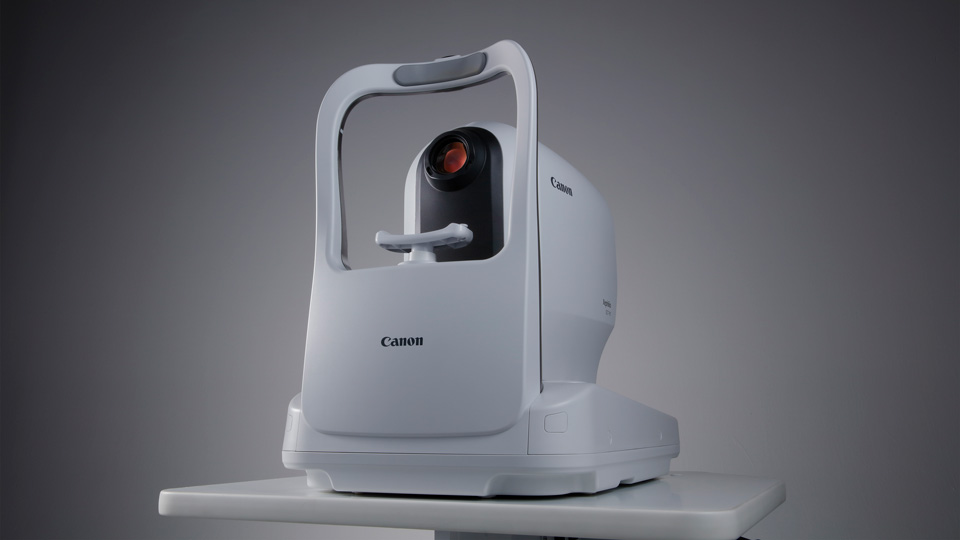
AI is already in the vast majority of our technology, from MRI and CT scans to eye care. We have products that can take non-invasive OCT-A images and make an average of 800 scans captured to produce the best scan possible, removing the noise and disturbance that might occur during the acquisition. But we want to take AI one step further.
What are the company’s aims for the next 12 months?
We are going through an expansion. With the new brand and investment in Canon eye care, we are also investing in headcount. We have just taken on Joe Blamire in the position of clinical support engineer.
We are hiring more clinical engineers. We find that a client in a hospital, screening site, or practice, may have a technical challenge, but when engineers arrive, they may also ask clinical questions that arise in their use of the device. We want our engineers to be able to answer both. We are bringing in people with clinical experience from NHS backgrounds, and then sending them through Canon’s in-house technical training programmes. This makes a unique hybrid of technical support staff who can seamlessly work between technical support and translation of clinical knowledge.
Beyond the brand
What trends do you see in the adoption of technology across both primary and secondary care?
Optometry and ophthalmology have to embrace cloud applications more. In the future, ophthalmology will need AI because eye diseases need to be monitored and managed over a longer period of time, because we are an ageing population, and we are living longer. In order to provide that level of service to the public, in the UK, we will need the support of technology like AI and medical devices with more smart technology.
The NHS as an institution has got to allow primary care to join that journey of change and evolution. Getting primary care connected into secondary care and providing secondary care with real-time information on the patient can only be done if the NHS adopts cloud-based applications and puts the training into primary care.
There has to be a clear framework that allows primary care in the local area to support it. The two sides are very different. One is very much an institution and is clinical. The other has some retail base, and some clinical base. There has to be a framework so that the two can integrate.
Optometrists are very skilled and can take further training to provide services. There are already models where optometrists are becoming glaucoma specialists or medical retina specialists. I think the ophthalmology community and optometry bodies need to sit together and ask: how can we put a framework together to allow for the transfer of real-time information?
High Street connectivity
I would like to commend High Street practices. They are investing in technology to differentiate and provide patients with cutting-edge clinical information. Optometry is making that investment and you see high-end technology going into practices. Some might even have dedicated diagnostic suites. Secondary care can benefit from that. Connectivity can bridge the two groups.
I think what we see is not the norm anymore. Everything is up for negotiation. The cost of logistics has gone up five-fold. The cost of manufacturing has gone up five-fold. I think globally companies are struggling with resource and if you don’t have the labour, you cannot produce the product, so what I’ve seen is a shift in the market in which the cost of goods to the end user has gone up. I think it’s elements of Brexit and elements of the international global supply chain, which means that the price of the end user product is increasing. That’s the challenge in our industry. I think people will need to be very selective when looking to adopt new technology to consider who will provide a complete value add. You might look at a product that does multiple things, or that is known to last a long time.

We are really excited about what the future of AI can bring about. We are also excited to grow the market with Canon. What makes me excited about this is that Canon has such a big brand presence globally. Most people can relate to Canon, the brand holds a lot of nostalgia for people, and it has a legacy behind it.
This autumn, OT is exploring the topic of ‘failing successfully’. What is your ethos?
I think what made Sense Medical a successful company is that we communicate a lot between colleagues, and we communicate effectively. In this day and age, we are much more under the spotlight of the Medicines and Healthcare Products Regulatory Agency.
When we started the company, we passed the certification of what was thought to be the strictest auditing company, TUV. I remember when the auditor came in, he said: ‘Is that all of you?’ For them, we were one of the first small companies they had worked with in the UK to have passed the audit, and they certified us as ISO 13485, which is used in the manufacturing of medical devices. We implemented it from day one but it wasn’t an easy journey – it took us two years, because the standard operating procedures have to be written. We have an internal auditor, and I am also a qualified auditor, so we can each audit different sections of the business. Then an external company comes in.
It is very hard, but because we have implemented such a high standard, there is a lot of risk management in place and so the ratio of failure is low.


Comments (0)
You must be logged in to join the discussion. Log in