- OT
- Industry
- Contact lenses
- Choosing carefully
Advertorial content is paid for and produced by a sponsor, and is reviewed and edited by the OT team before publication.
22 November 2017
In recent times, the contact lens industry has seen a revival in the soft frequent replacement category through the development of brand new materials, along with improvements to various existing designs, ensuring that eye care professionals (ECPs) have a lot more choice to offer their patients.
When choosing materials and lens care, careful consideration should be given to wearing patterns, environmental conditions and above all, compliance. As most ECPs recommend multi-purpose solutions now, it is vital that these lens care products can be efficacious and also enhance the wearing experience for the contact lens wearer.
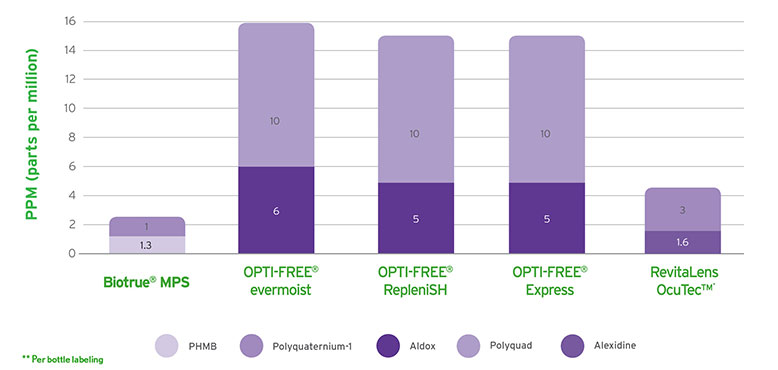
Biotrue® MPS has a dual-disinfecting system using polyaminopropyl biguanide (PHMB) and polyquaternium (PQ-1). This combination provides efficacy against bacteria, fungi, and Acanthamoeba.1,2 However, the specific pH value of Biotrue® MPS optimises the environment for these disinfectants meaning that not only do the log kill reductions for bacteria and fungi exceed the requirements for FDA/ISO stand-alone testing (see Figure 1),2 the concentrations of these preservatives are also extremely low (see Figure 2).
For extra comfort, at 7.5, the pH of Biotrue® MPS is designed to match the range of healthy tears (7.3-7.7)3 to help avoid any stinging reaction on lens application.
Figure 1: FDA/ISO stand-alone log kill requirements for disinfecting products
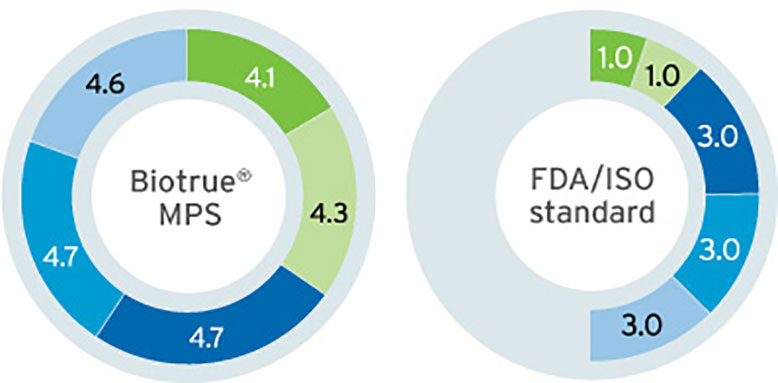
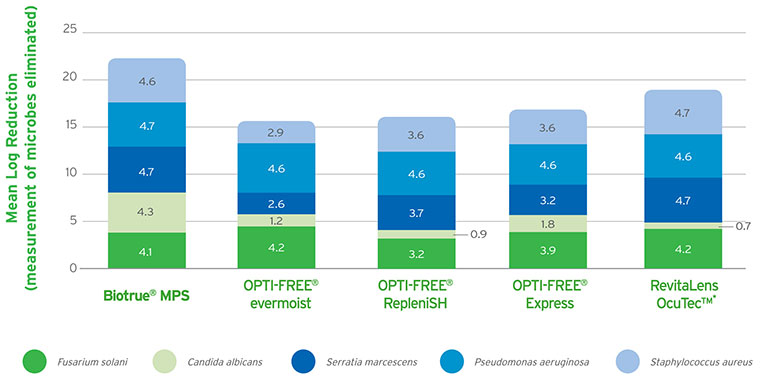
Figure 2: Overall disinfectant concentration**
Tears contain high concentrations of proteins, which have natural antibacterial properties. An example is lysozyme; in its active state, is directly toxic to bacteria (lysozyme hydrolyses the cell walls of bacteria).4
Biotrue® MPS contains an ingredient called sulfobetaine; a zwitterion, which surrounds and aids to protect active lysozyme, so it remains in the native state and retains its inherent activity.5 In fact, a study demonstrated that 90.1% of lysozyme was stabilised in its active form with Biotrue® MPS.6 Not all tear proteins are this useful. As they denature, proteins need to be eliminated to prevent deposition. Biotrue® MPS lifts and dissolves only denatured proteins, preventing them from building up as deposits on the contact lens surface.
Biotrue® MPS contains hyaluronan (HA). This hydrophilic glycosaminoglycan provides a moisture-rich hydrating network remaining on both hydrogel and silicone hydrogel lenses for up to 20 hours7 to keep lenses cushioned and hydrated for all day comfort.
Biotrue® MPS is an innovative solution. It has high biocidal efficacy yet is gentle on the surface of the eye, promotes the eye’s natural ability to defend itself from microbes by protecting active tear proteins and maintaining a long lasting hydrated surface on the contact lens. A solution for modern day reusable contact lenses.
References
- B. David, P. Walsh, S. Norton. (2010) Disinfection Efficacy of Multipurpose Solutions Against Acanthamoeba Using Methods Derived from the Current ISO 14729 Standards for Bacteria and Fungi. Bausch & Lomb Incorporated, Rochester, NY, USA
- Results of in vitro study following FDA/ISO stand-alone procedure for disinfecting products. Tests against all solutions were modified with organic soil added to create a more rigorous test condition. Primary criteria for effective disinfection are defined as a reduction in the number of bacteria by a minimum of 3 logs (99.9%) and a reduction of mold and yeast by a minimum of 1 log (90%) within the recommended disinfection time. Graphs depict mean log reduction measured after manufacturers’ recommended disinfecting time (soak only). Biotrue, OPTI-FREE RepleniSH, RevitaLens OcuTec, and OPTI-FREE evermoist results from separate in vitro studies performed following identical test procedures
- Burke.S; Fridman.K; Scheuer.C; Barniak.V; Physical Properties of Multipurpose Contact Lens Care Solutions
- Biological Magnetic Resonance data Bank; BMRB Featured system: Lysozyme
- Sindt.C; Review of Cornea and Contact Lenses; The Truth about Lysozyme; Oct 2010
- Vicki L. Barniak∗, Susan E. Burke, Srini Venkatesh. (2010) Comparative evaluation of multi-purpose solutions in the stabilization of tear lysozyme. Contact Lens & Anterior Eye
- Absorption and Controlled Release of a Polysaccharide Conditioning Agent from Soft Contact Lenses; Catherine A. Scheuer, Krista M. Fridman, Susan E. Burke, Srini Venkatesh.
* Outside of the USA, RevitaLens OcuTec is marked as Complete RevitaLens.


