- OT
- Industry
- Equipment and suppliers
- An “exquisitely detailed” picture
An “exquisitely detailed” picture
The eye health of cancer patients is being monitored using OCT

30 November 2017
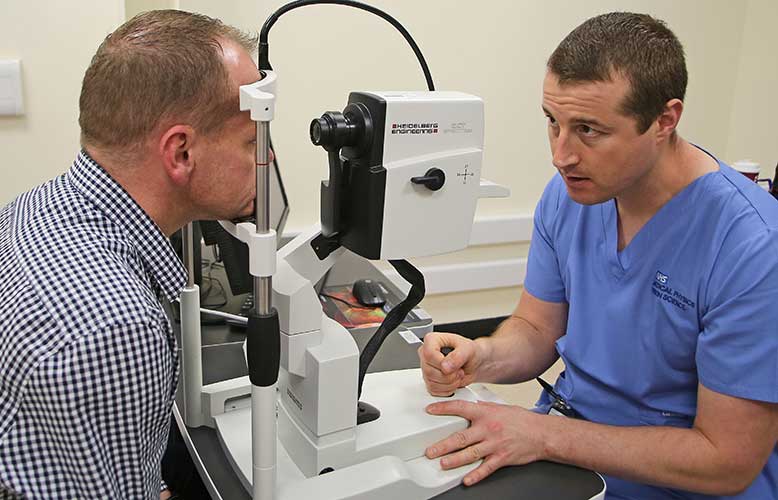
A piece of Heidelberg Engineering equipment is being used by the cancer care unit at the Royal Victoria Infirmary in Newcastle.
The Ophthalmology Clinic Trial Evaluation Suite has been assessing the eye health of cancer patients, with Heidelberg’s Spectralis optical coherence tomography (OCT) machine and a visual field analyser.
Patients are undergoing trials of small molecule anti-cancer drugs at the research centre, which consultant medical ophthalmologist, Dr Will Innes, said is a significant step forward in cancer treatment.
“With these drugs, the eye is ‘an organ at risk’ because some of the proteins and receptors that are targeted in cancer cells are also present in the eye. It is essential that we closely monitor eye health during treatment and, with this equipment, we can do that,” he explained.
The research centre is funded by the Sir Bobby Robson Foundation. The former England football team manager was treated by oncologist Professor Ruth Plummer, who is the director of the Sir Bobby Robson Cancer Research Trials Centre.
“Before we had this dedicated evaluation suite, patients had to visit separate areas of the hospital to have their eyes assessed – in some cases, on a weekly basis,” Dr Innes said.
“For anyone who is unwell, and especially for patients who are struggling to breathe because of cancer, it was very difficult to complete the eye checks necessary,” he added.
Dr Innes explained that OCT and another system that records electrical signals from the eye, optic nerve and brain, gives oncologists an “exquisitely detailed” picture of the eye’s structure and function. This allows oncologists to react quickly to changes during treatment.
“Oncologists are traditionally trained to deal with treatment side effects, such as hair loss, mouth and bowel problems and the dropping of white blood cell counts. With these new treatments ophthalmology is now also an essential element of monitoring,” Dr Innes said.
The centre enrolled over 600 new patients on clinical trials in the last year and is experiencing a year-on-year increase in the number of clinical trials open to recruitment.
Advertisement

Comments (0)
You must be logged in to join the discussion. Log in